Children’s Ibuprofen Recall
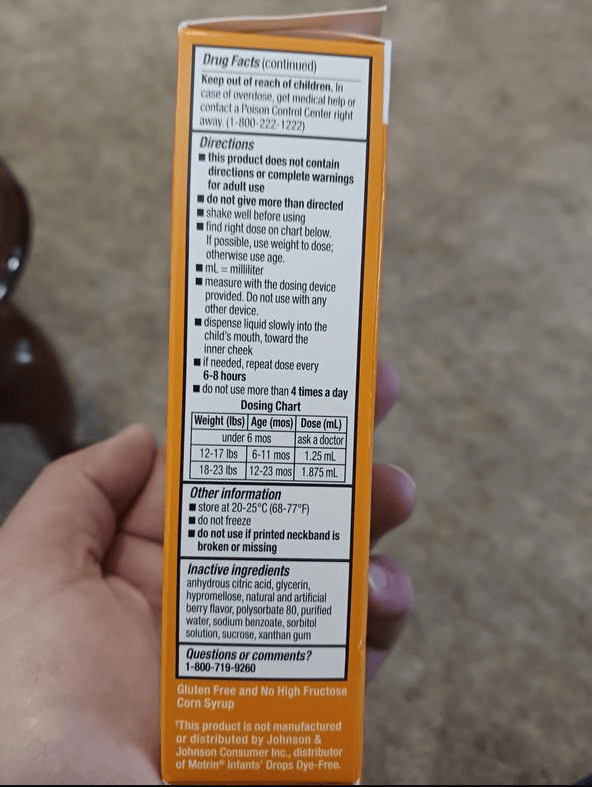
Almost 90,ooo bottles of children’s ibuprofen were recalled when a foreign substance was found in some, that could cause adverse health reactions. The recall was initiated by Strides Pharma on March 2 after reports of the product containing a “gel-like mass” and “black particles”. The exact product being recalled is the Children’s 4oz Ibuprofen Oral Suspension in Berry Flavor (a thick, liquid pain reliever). It was distributed by Taro Pharmaceuticals nationwide.
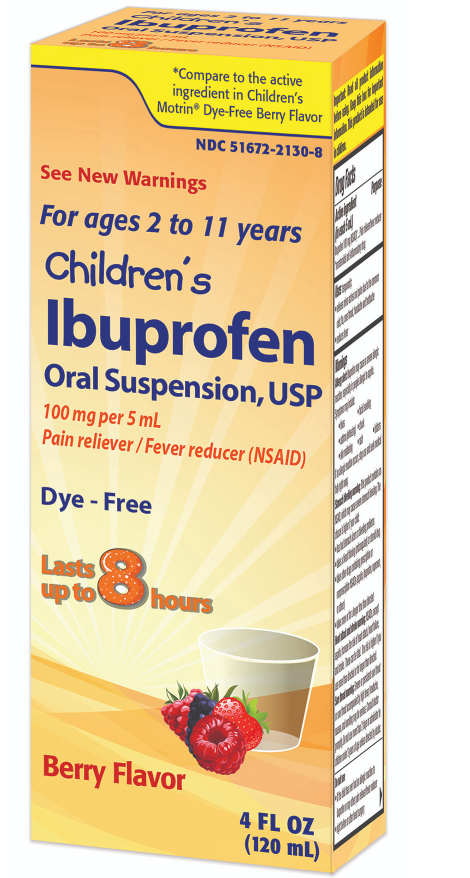
The recall affects 100-milligram Children’s Ibuprofen Oral Suspension, sold nationwide in a 4-fluid-ounce bottle. The lot numbers for the affected bottles are 7261973A and 7261974A, and the medication expires on Jan. 31, 2027, according to the FDA report.

- 7261973A
- 7261974A
- Expires on Jan. 31, 2027
Here’s What To Do If You Bought This Product:
- Check your medicine cabinet immediately
- Do not consume this product
- Get your money back – contact the store you purchased from.
- Discard of the product








Leave a Reply